Резюме
Остеопоротичні переломи зумовлюють серйозні витрати на охорону здоров’я, знижують незалежність/якість життя пацієнта і підвищують ризик смерті. Незважаючи на різні види терапії щодо зниження ризику майбутніх переломів, мало хто проходить лікування навіть після перелому стегна. Очевидно, що підходи минулого, спрямовані на зниження ризику остеопоротичних переломів, насамперед шляхом діагностики остеопорозу і призначення препаратів, що впливають на кісткову тканину, не були успішними. Потрібен інший підхід; таке зміщення фокуса й обговорюється в даній статті. Кажучи коротко, відповідно до концепції синдрому дисмобільності остеопоротичний перелом розглядають як клінічний результат наслідку і вважають, що остеопороз є лише частиною синдрому, який призводить до перелому, «пов’язаного з остеопорозом». Іншими компонентами цього синдрому є саркопенія, ожиріння, діабет, остеоартрит і теоретично безліч інших факторів, що збільшують ризик падінь із супутнім підвищеним ризиком перелому. Таким чином, концепція синдрому дисмобільності зрушує галузь дослідження, а також, що важливо, вивчення даних у літніх людей, у яких можливі остеопоротичні переломи, за межі єдиного фокуса на кістку для більш закономірного зосередження на цілісному підході до зниження ризику переломів.
Остеопоротические переломы обусловливают серьезные затраты на здравоохранение, снижают независимость/качество жизни пациента и повышают риск смерти. Несмотря на различные виды терапии по снижению риска будущих переломов, мало кто проходит лечение даже после перелома бедра. Очевидно, что подходы прошлого, направленные на снижение риска остеопоротических переломов, в первую очередь путем диагностики остеопороза и назначения препаратов, оказывающих влияние на костную ткань, не были успешными. Требуется другой подход; такое смещение фокуса и обсуждается в данной статье. Говоря коротко, в соответствии с концепцией синдрома дисмобильности остеопоротический перелом рассматривают как клинический исход последствия и полагают, что остеопороз является лишь частью синдрома, приводящего к перелому, «связанному с остеопорозом». Другими компонентами этого синдрома являются саркопения, ожирение, диабет, остеоартрит и теоретически множество других факторов, увеличивающих риск падений с сопутствующим повышенным риском перелома. Таким образом, концепция синдрома дисмобильности сдвигает область исследования, а также, что важно, изучение данных у пожилых людей, у которых возможны остеопоротические переломы, за пределы единственного фокуса на кость для более закономерного сосредоточения на целостном подходе к снижению риска переломов.
Fragility fractures engender major healthcare cost, reduce independence/quality and life and increase mortality risk. Despite availability of multiple therapies to reduce risk for future fracture, few patients are treated even following hip fracture. Clearly, approaches of the past aimed at reducing fragility fracture risk, primarily by diagnosing osteoporosis and initiating bone-active medications, have failed. A different approach is required; such a change in focus is proposed here. Briefly, the dysmobility syndrome concept recognizes fragility fracture as the clinical outcome of consequence and appreciates that osteoporosis is only part of the syndrome leading to “osteoporosis-related” fracture. Other components of this syndrome include sarcopenia, obesity, diabetes, osteoarthritis, and potentially multiple other factors that increase risk for falls with attendant increased fracture risk. In summary, the dysmobility syndrome concept moves the field, and also, importantly, older adults at risk for fragility fracture, beyond a singular focus on bone to more appropriately focus on a holistic approach to fracture risk reduction.
Scope of the Osteoporosis-Related Fracture Problem: Why Propose a New Syndrome?
Despite a worldwide increase in the older adult population, with concomitant increase in number of fragility fractures and their attendant morbidity, mortality and healthcare costs, fewer people are receiving medication to reduce fracture risk even following fracture [1, 2]. This failure to treat has recently been described as a “crisis” in osteoporosis care [3]. To address this osteoporosis treatment gap, international efforts have been, and are currently, focusing on the Fracture Liaison Service (FLS) model [4, 5]. The FLS paradigm consists of a systematic approach to identify those who have sustained a fragility fracture and subsequently execute appropriate evaluation to investigate secondary causes of bone loss and initiate pharmacologic therapy to reduce fracture risk [6]. The FLS approach has been widely studied and validated to reduce fractures and healthcare expenditures [1, 7].
To summarize, despite the availability of multiple rigo–rously studied medications that reduce fracture risk, and in the presence of an effective paradigm (FLS) to implement medical therapies, few people are being treated even after sustaining an osteoporosis-related fracture. In essence, the approaches previously taken to reduce fragility fracture risk, primarily by diagnosing low bone mass and initiating bone-active medications, have failed. Continuing to take the tactics of the past to inform clinicians and patients about the risks of “osteoporosis” seems similarly destined to fail; change is needed. This article suggests a different approach to the problem.
Dysmobility Syndrome: The Concept
It was recently advocated that the diagnosis of osteoporosis be expanded to include individuals at increased risk for fracture [8, 9]. At face value, this is a reasonable proposition as many older adults with osteopenia or even normal bone mineral density (BMD) sustain “osteoporosis-related” fractures [10]. Thus, diagnosing osteoporosis and treating only those with a BMD T-score ≤ –2.5 is not adequate to detect many people who will subsequently fracture. Conversely, some individuals with osteoporosis based upon BMD alone are at low fracture risk; clearly, moving beyond a BMD T-score based approach to guide fracture risk therapeutic initiation is appropriate. However, continued focus on a diagnosis of “osteoporosis”, or even on approaches for osteoporosis therapeutic intervention thresholds based upon estimated fracture risk, diverts attention from the
other concomitant problems contributing to fragility fracture risk.
The pathophysiology underpinning fragility fracture is often multifactorial and includes low bone mass and microarchitectural deterioration of bone leading to bone fragility [11] (i.e., in our opinion the CORRECT diagnosis of osteoporosis). It is osteoporosis, in concert with age-related loss of muscle mass and quality (i.e., sarcopenia), often combined with other age-related morbidities that negatively impact ambulation such as neuropathy, reduced ba–lance, impaired vision, polypharmacy, osteoarthritis and others leading to increased falls risk that ultimately causes the vast majority of “osteoporosis-related” fractures. That these age-related fragility fractures result from much more than simply compromised bone mass is exemplified by the fact that approximately one in six fragility fractures occur in those with normal proximal femur BMD [10]. Moreover, the multitude of rigorous prospective studies of effective bone-directed pharmacologic agents reproducibly demonstrate only an approximate 50–80 % reduction in vertebral fractures and an approximate 35 % reduction in non-vertebral fractures [12–16]. In summary, despite the best available therapies directed solely at improving bone, large number of fragility fractures continue to occur as other contributing factors lead to falls generating a force on the skeleton which can exceed the load-bearing capacity of even bone with normal mass.
It is apparent that factors independent of bone loss contribute to the age-related increase in fracture risk as fragility fractures increase dramatically with advancing age, but bone mass does not have a comparable decline [17]. A major contributor to this increase in fracture risk is falls. Falls become common with advancing age and approximately 95 % of hip fractures result from a fall [18]. Indeed, fall risk factors such as prior falls and slow gait speed predict hip fracture independent of BMD [19, 20]. Thus, it is unsurprising that poor physical performance is associated with increased hip fracture risk [21]. Such observations point to the importance of including sarcopenia as an integral consideration in efforts to reduce fragility fracture risk.
Sarcopenia has been defined as: “The age-associated loss of skeletal muscle mass and function… a complex syndrome associated with muscle mass loss alone or in conjunction with increased fat mass” [22]. This decline in muscle mass/function becomes common with advancing age [23] and is associated with impaired walking, falls and fractures. The pathogenesis of sarcopenia is complex but includes hormonal declines, increased inflammation, inadequate nutrition, sedentariness and toxin exposure [24]; factors well known to also cause osteoporosis. Indeed, it has been proposed that sarcopenia and osteoporosis are manifestations of the same processes in muscle and bone respectively [25]. Currently, sarcopenia is rarely diagnosed clinically, in large part reflecting the absence of a universally accepted diagnostic definition. To fill this void, a number of recent consensus conferences have proposed definitions, all of which include measurement of muscle mass and muscle function [22, 26, 27]. The recent definition proposed by the Foundation of the NIH Sarcopenia Project importantly integrates consideration of sarcopenic obesity (in essence, too much fat for the amount of muscle present) [28] by suggesting a unique approach to define low muscle mass as appendicular (leg + arm) lean mass divided by BMI [27]. Essentially, this is muscle mass/height corrected weight.
Considering sarcopenic obesity as a risk factor for fragi–lity fracture may seem counter-intuitive for some, as obesity has historically been thought of as being protective against fragility fractures by increasing mechanical load [29, 30]. However, that the relationship between fat and bone is much more complex than simple weight bearing is increasingly being recognized [31, 32]. To oversimplify, it is plausible that obesity leads to diversion of mesenchymal stem cells from osteoblasts to adipocytes thereby impairing bone formation. Moreover, infiltration of fat into muscle, or intramuscular adipose tissue (IMAT), leads to muscle dysfunction [33]. It is therefore not surprising that recent studies find obesity to increase fracture risk [34]. Clearly, consideration of obesity in fragility fracture risk estimation is appropriate.
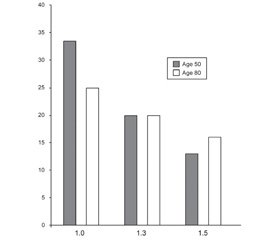
We believe that direct support for the importance of non-bone related factors in fracture risk is apparent given the inclusion of demographic, lifestyle and medical history factors included in fracture risk calculators such as FRAX and Garvan [35, 36]. Additionally, evidence that these calculators are not currently comprehensive and continue to evolve was recently provided by the implementation of a trabecular bone score (TBS) adjustment to the FRAX calculator. TBS is a bone assessment tool that is a surrogate for bone microarchitecture, independent of BMD [37], it’s effect on fracture risk calculation demonstrated that non-bone factors must be part of the cause for increased fracture risk with age. Specifically, in a study conducted by McCloskey et al. [38] lower TBS values exert a less profound effect on fracture risk in older individuals (fig. 1) indicating that bone fragility contributes less to fracture risk in more elderly people. A likely explanation for this observation is that falls and other age-related morbidities play a greater role while bone fragility plays a lesser role with advancing age.
/11-1.jpg)
Therefore, we believe that efforts to redefine osteoporosis as elevated fracture risk are off target. Instead, those interested in reducing fractures should focus on the multitude of diseases/conditions that lead to the ultimate adverse outcome of fragility fracture. Nonetheless, the “what’s in a name” publication [9] provide dincentive and direction to bring forward our suggestion that fragility fractures be considered part of a larger syndrome, that we suggested be named “dysmobility syndrome” and encompasses osteoporosis, sarcopenia, obesity and other fracture risk factors as an approach to improve identification and ultimately treatment of older adults to reduce their risk for falls and fractures [39]. The term “dysmobility”, i.e., impairment of or difficulty with walking, was selected, but it is acknowledged that other terminology for this concept could well be equally, or even more, descriptive of this concept. This approach is analogous to the widely recognized metabolic syndrome in which various conditions (e.g., hypertension, hyperlipidemia, etc.) are recognized contributors to the adverse outcome of cardiovascular disease (fig. 2 a, b).
/12-1.jpg)
To summarize, the dysmobility syndrome concept moves the field, and also, importantly, older adults at risk for fragility fracture, beyond a singular focus on bone. It seems reasonable that patients would accept such an approach to reduce their fracture risk similar to the approaches currently routinely taken to reduce risk following myocardial infarction. However, a critical factor will be education to inform the lay public that fragility fractures are often indicative of multi-system treatable problems, not simply “I fell”. Moreover, in our opinion, educational efforts are sorely needed that emphasize the adverse consequences related to fracture, specifically mortality, and likely of equal, or greater, importance, loss of independence that occurs following fragility fracture. The latter not only being devastating to a community dwelling individual, but also has huge societal impact due to the cost of long-term care.
In addition to recognizing this concept, approaches to the clinical identification of individuals with dysmobility syndrome will be required. As an initial approach to this, our group proposed [39] a simple score based system in which slow gait speed (< 1 m/sec), a BMD T-score ≤ –2.5, low appendicular lean mass as measured by DXA (4.45 and 7.26 kg/m2 for women and men respectively), history of falls within the last year, low grip strength (< 20 kg; female and < 30 kg; male) and high total body fat mass were considered with each risk factor receiving one point with dysmobility arbitrarily being diagnosed if the composite store was ≥ three.
It is certainly appropriate that fragility fracture risk factors other than these originally proposed be considered in the dysmobility syndrome concept. Two logical candidates include diabetes mellitus, and osteoarthritis. Diabetes is recognized to have adverse consequences upon bone and muscle [40, 41] and to increase fracture risk above that predicted by FRAX [42]. Similarly, some studies find osteoarthritis to be associated with increased risk for falls and fractures [43, 44]. Additionally, it is reasonable to consider use of narcotics for pain management or psychotropic medications as contributors to falls risk [45, 46]. Moreover, it is likely that factors considered for inclusion in dysmobility may not carry equal weight. Clearly, implementation of the dysmobility syndrome is a work in progress.
Nonetheless, we find the dysmobility syndrome concept to be a logical continuing evolution of the “osteoporosis” field. Notably, the field has moved beyond simple treatment based upon low BMD T-score to use of various calculators, e.g., FRAX, that were developed to improve detection of those at risk for fragility fracture. Such calculators clearly advance clinical decision-making, however, all are imperfect in that some individuals estimated to be at low risk sustain fractures [47]. Thus, improved approaches to identify at-risk individuals are needed; dysmobility syndrome seems a logical next step.
Is Dysmobility Syndrome Related to Adverse Health Outcomes?
The dysmobility syndrome concept is in its infancy. Nonetheless, an accumulating body of evidence finds dysmobility to be associated with adverse health outcomes. For example, in our initial editorial proposing the concept [39], we found dysmobility syndrome in 97 older adults, mean age 81 years, to be associated with a higher falls prevalence than proposed consensus sarcopenia definitions. Similarly, Edwards et al. in 398 older adults in the Herfordshire cohort found dysmobility to be associated prospectively with falls, but interestingly not with fractures [48]. Data from the MrOS study (reported only in abstract form) followed 5,826 older men for a mean of 6.2 years and found those with dysmobility to be more likely than those without to sustain major osteoporosis related fractures and hip fractures with hazard ratios of ~ 3 and ~ 2 respectively [49]. In this report, the diagnosis of dysmobility was an independent risk factor for fracture even after adjustment for FRAX score. Finally, Looker evaluated 2,975 NHANES participants age 50+ and found dysmobility to be associated with increased mortality risk, with highest hazard ratios (~ 3) for those with dysmobility who were 50–69 years of age [50]. In summary, existing, albeit preliminary, data find dysmobility syndrome to be associated with falls, fractures and mortality. Further optimization of the dysmobility syndrome approach is needed to evaluate whether some of the factors should receive greater weight and if additional factors should be included to enhance the capability to predict adverse health outcomes.
Can Dysmobility Syndrome be Implemented Clinically?
It seems theoretically possible that parameters of the dysmobility syndrome could be included into risk calculators, perhaps even integrated into FRAX. A hypothetical example of this concept is depicted in fig. 3. Large datasets and extensive analyses will be required before such calculators would be ready for clinical application.
/13-1.jpg)
In the interim, we believe that clinicians can implement the concept into clinical care while further evaluation is performed to optimize the diagnostic approach to dysmobility. To this end, it is logical that the dysmobility syndrome treatment paradigm follows, and expands upon, the approach used for “osteoporosis” today, i.e., recommendations for muscle strengthening exercise, optimization of nutrition (energy, calcium, vitamin D and protein), and bone active medications, i.e., our current “osteoporosis” medications. Moreover, it seems likely that a consensus definition for sarcopenia will be reached in the not too distant future, thus allowing clinicians to diagnose (and therefore treat) sarcopenia with non-pharmacologic measures. Additionally, it seems likely in the future that in addition to bone active medications, medications to increase muscle mass and function will be come available. Ideally, such new medications would improve both bone and muscle mass, and therefore physical function. Promising potential medications, including antibodies to myostatin [51] are in the research pipeline.
In conclusion, we find the dysmobility syndrome approach, (i.e., focusing on adverse health outcomes from falls and fractures, and not considering osteoporosis not as the single disease in need of diagnosis and treatment, but rather as simply a component of a risk profile) to be a logical extension of the “osteoporosis” treatment/fragility fracture prevention field. Moreover, it is the concept, not the name that is important; whether this is termed dysmobility syndrome or identified using other nomenclatureis irrelevant. Such a paradigm change seems likely to further remove the focus on a BMD T-score diagnosis, explain why many people sustain “osteoporosis-related” fractures with normal BMD, emphasize the inter-related nature of osteoporosis with sarcopenia and highlight the importance on considering the entire person, not simply one organ system, i.e., bone. In essence, this approach reminds us of the wise advice of William Osler who stated “The good physician treats the disease; the great physician treats the patient who has the disease” [52].
Conflicts of interests. Authors declare the absence of any conflicts of interests that might be construed to influence the results or interpretation of their manuscript.
Список литературы
1. Solomon DH, Johnston SS, Boytsov NN, McMorrow D, Lane JM, Krohn KD. Osteoporosis Medication Use after Hip Fracture in U.S. Patients between 2002 and 2011. J Bone Miner Res. 2014;29(9):1929-37. doi: 10.1002/jbmr.2202.
2. Kim SC, Kim DH, Mogun H. Impact of the U.S. Food and Drug Administration’s Safety-Related Announcements on the Use of Bisphosphonates After Hip Fracture. J Bone Miner Res. 2016;31(8):1536-40. doi: 10.1002/jbmr.2832.
3. Khosla S, Shane E. A Crisis in the Treatment of Osteoporosis. J Bone Miner Res. 2016;31(8):1485-7.doi: 10.1002/jbmr.2888.
4. Akesson K, Marsh D, Mitchell PJ, et al. Capture the Fracture: a Best Practice Framework and global campaign to break the fragility fracture cycle. Osteoporos Int. 2013;24(8):2135-52. doi: 10.1007/s00198-013-2348-z.
5. Eisman JA, Bogoch ER, Dell R, Harrington JT, et al. Making the first fracture the last fracture: ASBMR task force report on secondary fracture prevention. J Bone Miner Res. 2012;27(10):2039-46. doi: 10.1002/jbmr.1698.
6. Miller AN, Lake AF, Emory CL. Establishing a fracture liaison service: an orthopaedic approach. J Bone Joint Surg. 2015;97(8):675-81. doi: 10.2106/JBJS.N.00957.
7. Dell R, Greene D, Schelkun SR, Williams K. Osteoporosis disease management: the role of the orthopaedic surgeon. J Bone Joint Surg. 2008;90(suppl 4):188-194. doi: 10.2106/JBJS.H.00628.
8. Siris ES, Adler R, Bilezikian J, et al. The clinical diagnosis of osteoporosis: a position statement from the National Bone Health Alliance Working Group. Osteoporos Int. 2014;25(5):1439-43. doi: 10.1007/s00198-014-2655-z.
9. Siris ES, Boonen S, Mitchell PJ, Bilezikian J, Silverman S. What’s in a name? What constitutes the clinical diagnosis of osteoporosis? Osteoporos Int, 2012;23(8):2093-7. doi: 10.1007/s00198-012-1991-0.
10. Schuit SC, van der Klift M, Weel AE, et al. Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam study. Bone. 2004;34:195-202. PMID: 14751578.
11. Anonymous, Consensus Development Conference: Prophylaxis and Treatment of Osteoporosis. Am J Med. 1990;90:107-10. PMID: 1986575.
12. Black DM, Cummings SR, Karpf DB, et al. Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet. 1996;348(9041):1535-41. PMID: 8950879.
13. Black DM, Delmas PD, Eastell R, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356:1809-22. doi: 10.1056/NEJMoa067312.
14. McClung MR, Geusens P, Miller PD, et al. Effect of risedronate on the risk of hip fracture in elderly women. N Engl J Med. 2001;344:333-40. doi: 10.1056/NEJM200102013440503.
15. Lyles KW, Colón-Emeric CS, Magaziner JS, et al. Zoledronic acid and clinical fractures and mortality after hip fracture. N Engl J Med. 2007;357(18):1799-809. doi: 10.1056/NEJMoa074941.
16. Neer RM, Arnaud CD, Zanchetta JR, et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med. 2001;344(19):1434-41. doi: 10.1056/NEJM200105103441904.
17. Kelly TL, Wilson KE, Heymsfield SB. Dual energy X-Ray absorptiometry body composition reference values from NHANES. PLoS One. 2009;4(9):e7038. doi: 10.1371/journal.pone.0007038.
18. Parkkari J, Kannus P, Palvanen M, et al. Majority of hip fractures occur as a result of a fall and impact on the greater trochanter of the femur: a prospective controlled hip fracture study with 206 consecutive patients. Calcif Tissue Int. 1999;65(3):183-7. PMID: 10441647.
19. Dargent-Molina P, Favier F, Grandjean H, et al. Fall-related factors and risk of hip fracture: the EPIDOS prospective study. Lancet. 1996;348(9021):145-9. PMID: 8684153.
20. Nguyen DN, Pongchaiyakul C, Center JR, et al. Identification of high-risk individuals for hip fracture: A 14-year prospective study. J Bone Miner Res. 2005;20(11):1921-8. doi: 10.1359/JBMR.050520.
21. Cawthon PM, Fullman RL, Marshall L, et al. Physical performance and risk of hip fractures in older men. J Bone Miner Res. 2008;23(7):1037-44. doi: 10.1359/jbmr.080227.
22. Fielding RA, Vellas B, Evans WJ, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc. 2011;12(4):249-56. doi: 10.1016/j.jamda.2011.01.003.
23. Looker AC, Wang CY. Prevalence of reduced muscle strength in older U.S. adults: United States, 2011-2012. NCHS Data Brief. 2015;179:1-8. PMID: 25633238.
24. Morley JE, Malmstrom TK, Frailty, sarcopenia, and hormones. Endocrinol Metab Clin North Am. 2013;42(2):391-405. doi: 10.1016/j.ecl.2013.02.006.
25. Binkley N, Buehring B. Beyond FRAX: it’s time to consider “sarcoosteopenia”. J Clin Densitom. 2009;12(4):413-6. doi: 10.1016/j.jocd.2009.06.004.
26. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-23. doi: 10.1093/ageing/afq034.
27. Studenski SA, Peters KW, Alley DE, et al. The FNIH Sarcopenia Project: Rationale, Study Description, Conference Recommendations, and Final Estimates. J Gerontol A Biol Sci Med Sci. 2014;69(5):547-58. doi: 10.1093/gerona/glu010.
28. Prado CM, Wells JC, Smith SR, Stephan BC, Siervo M. Sarcopenic obesity: A Critical appraisal of the current evidence. Clin Nutr. 2012;31(5):583-601. doi: 10.1016/j.clnu.2012.06.010.
29. Cummings SR, Nevitt MC, Browner WS, et al. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N Engl J Med. 1995;332(12):767-73. doi: 10.1056/NEJM199503233321202.
30. Roy DK, O’Neill TW, Finn JD, et al. Determinants of incident vertebral fracture in men and women: results from the European Prospective Osteoporosis Study (EPOS). Osteoporos Int. 2003;14(1):19-26. doi: 10.1007/s00198-002-1317-8.
31. Rosen CJ, Bouxsein ML. Mechanisms of disease: is osteoporosis the obesity of bone. Nat Clin Pract Rheumatol. 2006;2(1):35-43. doi: 10.1038/ncprheum0070.
32. Rosen CJ, Klibanski A. Bone, fat and body composition: evolving concepts in the pathogenesis of osteoporosis. Am J Med. 2009;122:409-414. doi: 10.1016/j.amjmed.2008.11.027.
33. Addison O, Robin L. Marcus RL, LaStayo PC, Ryan AS. Intermuscular fat: a review of the consequences and causes. Int J Endocrinol. 2014;2014:309570. doi:10.1155/2014/309570.
34. Compston JE, Watts NB, Chapurlat R, et al. Obesity is not protective against fracture in postmenopausal women: GLOW. Am J Med. 2011;124:1043-50. doi: 10.1016/j.amjmed.2011.06.013.
35. Kanis JA, Burlet N, Cooper C, et al. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2008;19(4):399-428. doi: 10.1007/s00198-008-0560-z.
36. Nguyen ND, Frost SA, Center JR, Eisman JA, Nguyen TV. Development of prognostic nomograms for individualizing 5-year and 10-year fracture risks. Osteoporos Int. 2008;19(10):1431-44. doi: 10.1007/s00198-008-0588-0.
37. Silva BC, Leslie WD, Resch H, et al. Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res. 2014;29(3):518-30. doi: 10.1002/jbmr.2176.
38. McCloskey EV, Odén A, Harvey NC, et al. Adjusting fracture probability by trabecular bone score. Calcif Tissue Int. 2015;96(6):500-9. doi: 10.1007/s00223-015-9980-x.
39. Binkley N, Krueger D, Buehring B. What’s in a name revisited: should osteoporosis and sarcopenia be considered components of “dysmobility syndrome”? Osteoporos Int. 2013;24(12):2955-9. doi: 10.1007/s00198-013-2427-1.
40. Schwartz AV. Epidemiology of fractures in type 2 diabetes. Bone. 2016;82:2-8. doi: 10.1016/j.bone.2015.05.032.
41. Wang J, You W, Jing Z, Wang R, Fu Z, Wang Y. Increased risk of vertebral fracture in patients with diabetes: a meta-analysis of cohort studies. Int Orthop. 2016;40(6):1299-307. doi: 10.1007/s00264-016-3146-y.
42. Giangregorio LM, Leslie WD, Lix LM, et al. FRAX underestimates fracture risk in patients with diabetes. J Bone Miner Res. 2012;27(2):301-8. doi: 10.1002/jbmr.556.
43. Prieto-Alhambra D, Nogues X, Javaid MJ, et al. An increased rate of falling leads to a rise in fracture risk in postmenopausal women with self-reported osteoarthritis: a prospective multinational cohort study (GLOW). Ann Rheum Dis. 2013;72(6):911-7. doi: 10.1136/annrheumdis-2012-201451.
44. Zasadzka E, Borowicz AM, Roszak M, Pawlaczyk M. Assessment of the risk of falling with the use of timed up and go test in the elderly with lower extremity osteoarthritis. Clin Interv Aging. 2015;10:1289-98. doi: 10.2147/CIA.S86001.
45. Ebly EM, Hogan DB, Fung TS. Potential adverse outcomes of psychotropic and narcotic drug use in Canadian seniors. J Clin Epidemiol. 1997;50(7):857-63. PMID: 9253399.
46. Rolita L, Spegman A, Tang X, Cronstein BN. Greater number of narcotic analgesic prescriptions for osteoarthritis is associated with falls and fractures in elderly adults. J Am Geriatr Soc. 2013;61(3):335-40. doi: 10.1111/jgs.12148.
47. Bolland MJ, Siu AT, Mason BH, et al. Evaluation of the FRAX and Garvan fracture risk calculators in older women. J Bone Miner Res. 2011;26:420-7. doi: 10.1002/jbmr.215.
48. Clynes MA, Edwards MH, Buehring B, Dennison EM, Binkley N, Cooper C. Definitions of Sarcopenia: Associations with Previous Falls and Fracture in a Population Sample. Calcif Tissue Int. 2015;97(5):445-52. doi: 10.1007/s00223-015-0044-z.
49. Buehring B, Lewis B, Hansen K, et al. Association of Musculoskeletal Health Score with Fracture Risk in the MrOS cohort. J Bone Miner Res. 2016;31(Suppl 1). Available from: http://www.asbmr.org/education/AbstractDetail?aid=104ce55b-45f8-4bcb-9b01-402428abafe4.
50. Looker AC. Dysmobility syndrome and mortality risk in US men and women age 50 years and older. Osteoporos Int. 2015;26(1):93-102. doi: 10.1007/s00198-014-2904-1.
51. Buehring B, Binkley N. Myostatin — the holy grail for muscle, bone, and fat? Curr Osteoporos Rep. 2013;11(4):407-14. doi: 10.1007/s11914-013-0160-5.
52. Osler W. Sir William Osler. Aphorisms, ed. W.B. Bean. 1961, Springfield IL: C.C. Thomas, Oxford, Blackwell Scientific Publs.

/11-1.jpg)
/12-1.jpg)
/13-1.jpg)