Резюме
Актуальність. Низькотравматичні переломи є важливими ускладненнями системного остеопорозу, що призводять до зниження якості життя, збільшення захворюваності, інвалідності та смертності. У наш час мінеральна щільність кісткової тканини (МЩКТ) і трабекулярний кістковий індекс (ТКI), що визначаються за допомогою двохенергетичної рентгенівської абсорбцiометрiї, є двома основними параметрами міцності кістки. Вони широко використовуються при діагностиці остеопорозу. Метою дослідження було вивчення частоти і локалізації різних остеопоротичних переломів у жінок в постменопаузальному періоді залежно від віку, МЩКТ і ТКI. Матеріали та методи. Проведено одномоментне дослідження і проаналізовані дані 1369 жінок у постменопаузальному періоді (вік 45–89 років). Пацієнтки були розділені на групи залежно від МЩКТ згідно з критеріями Всесвітньої організації охорони здоров’я: нормальний стан кісткової тканини (T-індекс > –1,0 стандартного відхилення (СВ)), остеопенія (≤ –1,0 T-індекс > –2,5 СВ), остеопороз (T-індекс ≤ –2,5 СВ), і в залежності від квартилів (груп) ТКI (L1-L4): QI — найнижча квартиль (0,36–1,08); QII — нижня квартиль (1,08–1,20); QIII — верхня квартиль (1,20–1,32); QIV — найвища квартиль (1,32–1,79). МЩКТ і ТКI поперекового відділу хребта (L1-L4) вимірювали за допомогою денситометра Prodigy (GE Medical systems, Lunar, модель 8743, 2005). ТКI визначали з використанням програмного забезпечення TBS Insight (Med-Imaps, Bordeaux, France, 2006). Результати. Встановлено, що ризик усіх типів остеопоротичних переломів у жінок достовірно збільшується з віком (вертебральних переломів — у 1,97–4,26 раза, невертебральних — у 1,96–5,45, комбінованих — у 1,54—3,14 раза). Крім того, ми виявили, що остеопоротичні переломи присутні у всіх групах жінок iз різною МЩКТ: при остеопорозі (45,5 %), остеопенiї (26,9 %) та за нормального стану кісткової тканини (18,1 %). Найбільшою частота остеопоротичних переломів була при остеопорозі (46,5 %), переважно в жінок iз хребцевими і комбінованими переломами. Тільки в 25,5 % пацієнток iз невертебральними переломами спостерігається остеопороз. Низький ТКI відзначався в 50,8 % жінок iз вертебральними, 43,7 % — невертебральними і в 32,8 % — iз комбінованими (хребцевими і невертебральними) остеопоротичними переломами. Висновки. Остеопоротичні переломи частково пов’язані з віком, МЩКТ і ТКI. Однак існують й інші важливі чинники, що впливають на ризик переломів, і їх поєднання вимагає подальшого вивчення.
Актуальность. Низкотравматичные переломы являются важными осложнениями системного остеопороза, которые приводят к снижению качества жизни, увеличению заболеваемости, инвалидности и смертности. В настоящее время минеральная плотность костной ткани (МПКТ) и трабекулярный костный индекс (ТКИ), определяемые посредством двухэнергетической рентгеновской абсорбциометрии, являются двумя основными параметрами прочности кости. Они широко используются при диагностике остеопороза. Целью исследования было изучение частоты и локализации различных остеопоротических переломов у женщин в постменопаузальном периоде в зависимости от возраста, МПКТ и ТКИ. Материалы и методы. Проведено одномоментное исследование и проанализированы данные 1369 женщин в постменопаузальном периоде (возраст 45–89 лет). Пациентки были разделены на группы в зависимости от МПКТ согласно критериям Всемирной организации здравоохранения: нормальное состояние костной ткани (T-индекс > –1,0 стандартного отклонения (СО)), остеопения (≤ –1,0 T-индекс > –2,5 СО), остеопороз (T-индекс ≤ –2,5 СО), и в зависимости от квартилей (групп) ТКИ (L1-L4): QI — самая низкая (0,36–1,08); QII — нижняя квартиль (1,08–1,20); QIII — верхняя квартиль (1,20–1,32); QIV — самая высокая квартиль (1,32–1,79). МПКТ и ТКИ поясничного отдела позвоночника (L1-L4) измеряли с помощью денситометра Prodigy (GE Medical systems, Lunar, модель 8743, 2005). ТКИ определяли с использованием программного обеспечения TBS Insight (Med-Imaps, Bordeaux, France, 2006). Результаты. Установлено, что риск всех типов остеопоротических переломов у женщин достоверно увеличивается с возрастом (вертебральных переломов — в 1,97–4,26 раза, невертебральных — в 1,96–5,45, комбинированных — в 1,54–3,14 раза). Кроме того, мы обнаружили, что остеопоротические переломы присутствуют во всех группах женщин с различной МПКТ: при остеопорозе (45,5 %), остеопении (26,9 %) и нормальном состоянии костной ткани (18,1 %). Наибольшей частота остеопоротических переломов была при остеопорозе (46,5 %), преимущественно у женщин с вертебральными и комбинированными переломами. Только у 25,5 % пациенток с невертебральными переломами наблюдается остеопороз. Низкий ТКИ отмечался у 50,8 % женщин с вертебральными, 43,7 % — невертебральными и у 32,8 % — с комбинированными (вертебральными и невертебральными) остеопоротическими переломами. Выводы. Остеопоротические переломы частично связаны с возрастом, МПКТ и ТКИ. Однако существуют и другие важные факторы, влияющие на риск переломов, и их сочетание требует дальнейшего изучения.
Background. Low-trauma fractures are the important complications of systemic osteoporosis, which lead to reduced quality of life, increased morbidity, disability and mortality. Nowadays, bone mineral density (BMD) and trabecular bone score (TBS) measured by dual energy X-ray absorptiometry are two major parameters of bone strength. They are widely used when establishing the osteoporosis diagnosis. The purpose was to study the frequency and localization of different osteoporotic fractures in postmenopausal women depending on the age, BMD and TBS. Materials and methods. We have performed cross-sectional study and have examined 1,369 postmenopausal women aged 45–89 years. Patients were divided into groups depending on BMD according to the WHO criteria: normal bone (T-score > –1.0 standard deviation (SD)), osteopenia (≤ –1.0 T-score ≤ –2.5 SD), osteoporosis (T-score ≤ –2.5 SD), and depending on TBS (L1-L4) quartiles (groups): QI — the lowest quartile (0.36–1.08); QII — the lower quartile (1.08–1.20); QIII — the upper quartile (1.20–1.32); QIV — the highest quartile (1.32–1.79). BMD and TBS in lumbar spine (L1-L4) were measured using Prodigy densitometer (GE Medical systems, Lunar, model 8743, 2005). TBS was determined using the software TBS Insight (Med-Imaps, Bordeaux, France, 2006). Results. Our study found that risk of all types of osteoporotic fractures in females reliably increases with age (for vertebral fractures — by 1.97–4.26 times, non-vertebral — 1.96–5.45 times, combined fractures — 1.54–3.14 times). Additionally, we have revealed that osteoporotic fractures are present in all groups of females with different BMD: osteoporosis (45.5 %), osteopenia (26.9 %) and normal bone (18.1 %). The highest frequency of osteoporotic fractures was detected in osteoporosis (46.5 %), and, predominantly, in women with vertebral and combined fractures. Only 25.5 % of patients with non-vertebral fractures have osteoporosis. Low TBS was observed in 50.8 % of females with vertebral, 43.7 % — non-vertebral and 32.8 % — with combined (vertebral and non-vertebral) osteoporotic fractures. Conclusions. Osteoporotic fractures are partly associated with age, BMD and TBS. However, there are other significant factors, which influence the risk of fractures, and their combination requires further study.
Introduction
Osteoporotic fractures are tremendous medico-social problem in almost all industrialized countries [1–6]. Every year, up to 9 millions of new low-trauma fractures caused by osteoporosis are expected [3]. The rate of mortality associated with fractures of the hip and spine can exceed 20 % [3]. In the United States, about 2 million new fractures are registered each year [7]. Approximately 10 % of 50-year-old women already have at least one osteoporotic fracture [1, 5]. Given the steady increase of life expectancy, these figures will be doubled in the next 40–50 years [8].
Current studies confirm that dual-energy X-ray absorptiometry (DXA) is the gold standard for diagnosing osteoporosis and its indices (bone mineral density (BMD) and trabecular bone score (TBS) are the important parameters of bone density, microarchitecture and strength). However, their relationship with the fractures of different locations (vertebral or non-vertebral) continues to be studied, existing data are limited and depend on age, sex and some national features.
The purpose of this research was to evaluate the frequency and localization of different osteoporotic fractures in postmenopausal women in Ukrainian cohort depending on BMD and TBS.
Materials and methods
Participants. We have performed a cross-sectional, case-control research. The study was conducted in Ukrainian Scientific Medical Center of Osteoporosis and department of clinical physiology and pathology of locomotor apparatus of D.F. Chebotarev Institute of Gerontology of the NAMS of Ukraine. It was approved by ethics committee of the institute (12/10/2013). All patients signed informed consent for participation in the study. 1,369 postmenopausal women aged 45–89 years were examined (mean age — 62.5 [55.0; 73.0] years, mean body mass index — 28.6 ± 6.5 kg/m2, the average duration of postmenopausal period — 14.5 ± 9.5 years). We excluded the patients with severe concomitant diseases, high-energy fractures or trauma history, high levels of physical activity, secondary osteoporosis or conditions that could affect the bone, some significant disorders (connective tissue disease, neoplasms in the past medical history, administration of corticosteroids, alcohol abuse and others), clinically manifested vertebral fractures with post-fracture period duration at least 6 months.
For analysis, we divided our patients into groups according to age, BMD and TBS indices. At first, they were divided into three age groups (45–59, 60–74 and 75–89 years). In addition, they were divided depending on BMD parameters according to the World Health Organization criteria and International Society for Clinical Densito–metry guidelines. The assessment of BMD indexes was performed according to next DXA criteria: normal bone (T-score > –1.0 standard deviation (SD)), osteopenia (≤ –1.0 T-score > –2.5 SD) and osteoporosis (T-score ≤ –2.5 SD) [5, 9]. The women were divided into next groups: first one (NB) — 563 females with normal BMD parameters, se–cond one (OPEN) — 498 women with osteopenia, third one (OP) — 308 patients with osteoporosis.
Subsequently, patients were divided depending on TBS. Nowadays, TBS has no standard indicators and in various countries, including Ukraine, studies to determine its refe–rence indices are only in process [10]. Standard TBS, as well as BMD, should be specific for definite geographic areas and ethnic groups. Therefore, this section describes a statistical approach that is used in cases of irregular quantitative values, namely quartile distribution groups.
All examined females were divided according to TBS parameters (L1-L4) into groups (quartiles): QI — the lowest quartile (0.36–1.08; n = 152); QII — the lower quartile (1.08–1.20; n = 156); QIII — the upper quartile (1.20–1.32; n = 148); QIV — the highest quartile (1.32–1.79; n = 133).
Presence, localization, mechanism and time of fracture were determined using specialized questionnaire developed in Ukrainian Scientific Medical Center of Osteoporosis. All information was gathered from patients by interview. In present study, we evaluated the frequency of vertebral, non-vertebral (non-spine non-hip fractures: humerus, forearm, clavicle, ribs, pelvis, upper and lower extremities) and combined fractures.
Methods. Bone mineral density and TBS were measured at the lumbar spine (L1-L4) using dual energy X-ray Prodigy densitometer (GE Medical systems, Lunar, model 8743, 2005). Trabecular bone score was determined using TBS Insight software (Med-Imaps, Bordeaux, France, 2006).
Statistical analysis was performed using Statistica 10.0 software package (StatSoft© Inc., 1984–2011). All variables were distributed using the Kolmogorov-Smirnov test. Intergroup comparisons were made using one-way analysis of variance and Mann-Whitney U test. Differences in the distribution of samples were assessed using χ2 test. To evaluate the correlation between variables, we used Spearman’s rank correlation coefficient (R). The critical level of significance was p < 0.05.
Results
The results of our study demonstrated that age is the significant risk factor for osteoporotic fractures in females. Fracture distribution analysis according to the age has shown that frequency of low-trauma osteoporotic fractures (vertebral, non-vertebral and combined) progressively increased from 22.2 % in women aged 45–59 years to 35.8 % in patients aged 75–89 years (Fig. 1). The most significant increase was determined for vertebral fractures.
In addition, it was found that osteoporotic fractures were present in all groups of patients with different BMD: osteoporosis — 45.5 %, osteopenia — 26.9 % and normal bone — 18.1 %. Moreover, the frequency of non-vertebral low-trauma fractures was not significantly different in women with osteoporosis (11.7 %), osteopenia (11.0 %) and normal BMD (9.8 %).
Also, we observed significant differences (χ2 = 95.66, p < 0.0001) due to vertebral (OP-group — 20.5 %, OPEN-group — 9.6 %, NB-group — 4.3 %) and combined fractures (OP-group — 13.3 %, OPEN-group — 6.2 %, NB-group — 5.0 %). However, the frequency of combined fractures (vertebral and non-vertebral) in OPEN- and NB-group did not differ significantly (Fig. 2).
It was found that frequency of osteoporosis (46.6 %) was significantly higher among patients with vertebral fractures compared to females with normal BMD (p = 0.02). Additionally, 53.4 % of women with vertebral fractures had normal BMD (17.8 %) or osteopenic syndrome (35.6 %).
Moreover, patients with osteoporosis accounted for about a quarter (25.5 %) of 141 women with non-vertebral fractures (OPEN-group — 39.0 %, NB-group — 35.5 %). The number of females with osteoporosis who had combine (vertebral and non-vertebral) fractures was 41.0 % and was not significantly different from rates of patients with osteopenia (31.0 %) and normal BMD (28.0 %) (Fig. 3).
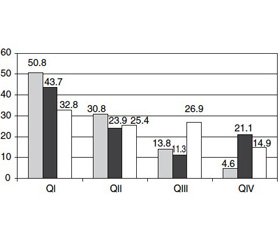
Furthermore, we have studied the fractures distribution in postmenopausal women depending on TBS. Our study revealed that despite the lowest TBS (QI), only 50.8 % of vertebral, 43.7 % of non-vertebral and 32.8 % of combined fractures occurred (Fig. 4).
The results of correlation analysis showed the significant association between the presence of any fracture and age (R = 0.21), BMD (R = –0.28) and TBS (R = –0.26). We also found the significant correlation between TBS and age (highest in NB-group: OP-group — –0.26 (p < 0.05); OPEN-group — –0.33 (p < 0.05); NB-group — –0.43 (p < 0.05)), presence of fractures (highest in OPEN-group: OP-group — –0.23 (p < 0.05); OPEN-group — –0.32 (p < 0.05); NB-group — –0.16 (p < 0.05)), and bone mi–neral density (only in OPEN-group: 0.23 (p < 0.05)).
Discussion
It is well known that systemic osteoporosis is an important skeletal disease characterized by low bone mass and microarchitectural deterioration of bone with increased bone fragility and risk of fractures [1, 3, 5]. Low-trauma fractures are the significant complications of systemic osteoporosis, which lead to reduced quality of life, an increase of morbi–dity, disability and mortality. The frequency of vertebral and non-vertebral fractures (hip, proximal humerus, distal forearm, etc.) progressively increases in males and females with age, however, it has sex and national features.
Nowadays, the DXA with BMD is gold standard for osteoporosis diagnosis in the absence of detected low-trauma fractures [3]. BMD is one of the main factors that determine bone strength and fracture risk [11], however, there is a significant overlap of BMD indexes among people with and without fractures [12]. There are many other factors that have influence on bone strength and fracture risk, such as macro- and microarchitecture of the bone, presence of microdamages, mineralization of bone matrix and remodeling speed [13, 14]. In recent years, new methods to assess bone microarchitecture were deve–loped. Among non-invasive methods, which significantly improve imaging techniques, there are quantitative (peripheral) computed tomography and magnetic resonance imaging, which allow the direct measurement of bone microarchitecture [5]. However, both methods are impractical for routine screening and monitoring of treatment [15].
According to DXA, the main quantitative characteristic of bone is BMD, the main qualitative parameter — trabecular bone score. It is known that both indices are decreasing with age, worsening the state of bone [12, 16–19]. It depends on the mean thickness and volume fraction of trabecular bone microarchitecture. Significant correlations between TBS and 3D-parameters of bone microarchitecture in corpses were found earlier. The largest correlation was between TBS and the density of connections that explained 67.2 % of variance. Higher TBS reflected better bone strength characteristics, while lower values showed weakness of bone and susceptibility to fractures [12, 19]. TBS may be determined retrospectively based on the DXA images made in advance without the need for any additional studies. Moreover, the rate can be compared to BMD as it determines bone state in the same region. A number of informative studies on simultaneous use of TBS and BMD were conducted [13, 14, 20]. It was proved that in postmenopausal women who had a history of fractures, the index is less than in individuals of corresponding age without fractures [17, 18, 20].
The aim of this study was to evaluate the frequency of osteoporotic fractures in postmenopausal women depending on BMD and TBS.
Our results show that age is the significant risk factor for all osteoporotic fractures. Frequency of low-trauma fractures (vertebral, non-vertebral and combined) progressively increased from 22.2 % in women aged 45–59 years to 35.8 % in patients aged 75–89 years.
In addition, it was found that osteoporotic fractures are present in all examined groups of women with different BMD: osteoporosis — 45.5 %, osteopenia — 26.9 % and normal bone — 18.1 %. Moreover, the frequency of non-vertebral fractures was not significantly different in women with osteoporosis (11.7 %), osteopenia (11.0 %) and normal BMD (9.8 %). Significant differences (χ2 = 95.66, p < 0.0001) were observed due to vertebral (OP-group — 20.5 %, OPEN-group — 9.6 %, NB-group — 4.3 %) and combined fractures (OP-group — 13.3 %, OPEN-group — 6.2 %, NB-group — 5.0 %).
Among patients with vertebral fractures, number of persons with osteoporosis (46.6 %) was significantly higher compared to those with normal BMD (p = 0.02). Additionally, 53.4 % of women with vertebral fractures had normal BMD (17.8 %) or osteopenic syndrome (35.6 %).
It was found that patients with osteoporosis accounted for about a quarter (25.5 %) among women with non-vertebral fractures (OPEN-group — 39.0 %, NB-group — 35.5 %). Moreover, the proportion of patients with osteoporosis who had combine (vertebral and non-vertebral) fractures was 41.0 % and was not significantly different from rates of patients with osteopenia (31.0 %) and normal BMD (28.0 %).
Current literature data confirmed the significantly lower spine TBS and BMD in women aged 50 years or older with major osteoporotic, spine and hip fractures (all p < 0.0001). Spine TBS and BMD predicted fractures equally well, and the combination was reliable superior to either measurement alone. Spine TBS predicts osteoporotic fractures and provides information that is independent of spine and hip BMD. Combining TBS with BMD incrementally improved fracture prediction in postmenopausal women. Multiple linear regression and logistic regression (lowest vs highest tertile) was used to define the sensitivity of TBS to other risk factors associated with osteoporosis. Only a small number of the TBS measurement (7–11 %) could be explained by BMD. In multiple linear regression and logistic regression models, reduced lumbar spine TBS was associated with recent major fracture, rheumatoid arthritis, chronic obstructive pulmonary disease, high alcohol intake, and higher body mass index [17].
Our assessment of fracture types (vertebral, non-vertebral or combined) in women depending on TBS showed that despite the lowest TBS (QI), only 50.8 % of vertebral, 43.7 % of non-vertebral and 32.8 % of combined fractures occurred. We also found the significant correlation between TBS and age (highest in NB-group), presence of fractures (highest in OPEN-group) and bone mineral density (only in OPEN-group).
Conclusions
Our study confirmed an increase of low-trauma fractures in postmenopausal women with age and showed the highest frequency of osteoporotic fractures in patients with osteoporosis (45.5 %). The proportion of patients with osteoporosis among females with non-vertebral fractures was 25.5 %. Moreover, 50.8 % of vertebral, 43.7 % of non-vertebral and 32.8 % of combined fractures occur on a background of poor quality of trabecular bone. The presence of low-trauma fractures is partly associated with age, bone mineral density and trabecular bone score. However, there are other significant factors influencing the risk of fractures. The future studies on BMD and TBS measurement toge–ther with other risk factors are needed.
Acknowledgements. The authors would like to acknow–ledge the assistance of all the members of the department of clinical physiology and pathology of locomotor apparatus of D.F. Chebotarev Institute of Gerontology of the NAMS of Ukraine (Kyiv) for DXA measurement.
Conflicts of interest. The authors report no conflicts of interest.
Funding statement. No sponsors funded nor had any decision in the development, review or submission of this article.
Список литературы
1. Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res. 2007 Mar;22(3):
465-75. doi: 10.1359/jbmr.061113.
2. Curtis EM, Moon RJ, Harvey NC, Cooper C. Reprint of: The impact of fragility fracture and approaches to osteoporosis risk assessment worldwide. Int J Orthop Trauma Nurs. 2017 Aug;26:7-17. doi: 10.1016/j.ijotn.2017.04.004.
3. Davies KM, Stegman MR, Heaney RP, Recker RR. Prevalence and severity of vertebral fracture: The Saunders County Bone Quality Study. Osteoporos Int. 1996;6(2):
160-5.
4. Fischer S, Kapinos KA, Mulcahy A, Pinto L, Hayden O, Barron R. Estimating the long-term functional burden of osteoporosis-related fractures. Osteoporos Int. 2017 Oct;28(10):2843-2851. doi: 10.1007/s00198-017-4110-4.
5. Povorozyuk VV, Grygorieva NV, Orlyk TV, et al. –Osteoporosis in the practice of an internist-doctor. Кyiv; 2014. 198 p. (in Russian).
6. Czerwinski E, Badurski JE, Marcinowska-Suchowierska E, Osieleniac J. Current understanding of osteoporosis according to the position of the World Health Organization (WHO) and International Osteoporosis Foundation. Ortop Traumatol Rehabil. 2007 Jul-Aug;9(4):337-56.
7. Leslie WD, Lix LM, Langsetmo L, et al. Construction of a FRAX® model for the assessment of fracture probability in Canada and implications for treatment. Osteoporos Int. 2011 Mar;22(3):817-27. doi: 10.1007/s00198-010-1464-2.
8. Johnnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. 2006 Dec;17(12):1726-33. doi: 10.1007/s00198-006-0172-4.
9. ISCD. Official Positions 2015 ISCD - Adult and Pediatric. Available from: https://iscd.app.box.com/v/
OP-ISCD-2015-Adult/. Accessed June 30, 2018.
10. Povoroznyuk VV, Dzerovich NI, Orlyk TV. Trabecular Bone Score in Clinical Practice: Review of Literature and Results of Own Research. Problem of osteology 2014;(17): 3-13. (in Ukrainian).
11. Johnell O, Kanis JA, Oden E, et al. Predictive value of BMD for hip and other fractures. J Bone Miner Res. 2005 Jul;20(7):1185-94. doi: 10.1359/JBMR.050304.
12. Hans D, Barthe N, Boutroy S, Pothuaud L, Winzenrieth R, Krieg MA. Correlations between TBS, measured using antero-posterior DXA acquisition, and 3D parameters of bone micro-architecture: an experimental study on human cadavre vertebrae. J Clin Densitom. 2011 Jul-Sep;14(3):302-12. doi: 10.1016/j.jocd.2011.05.005.
13. Leslie WD, Pahlavan PS, Tsang JF, Lix LM; Manitoba Bone Density Program. Prediction of hip and other osteoporotic fractures from hip geometry in a large clinical cohort. Osteoporos Int. 2009 Oct;20(10):1767-74. doi: 10.1007/s00198-009-0874-5.
14. Rubin CD. Emerging concepts in osteoporosis and bone strength. Curr Med Res Opin. 2005 Jul;21(7):1049-56. doi: 10.1185/030079905X50525.
15. Griffith JF, Engelke K, Genant HK. Looking Beyond bone mineral density: imaging assessment of bone quality. Ann N Y Acad Sci. 2010 Mar;1192:45-56. doi: 10.1111/j.1749-6632.2009.05378.x.
16. Harvey NC, Glüer CC, Binkley N, et al. Trabecular bone score (TBS) as a new complementary approach for osteoporosis evaluation in clinical practice. Bone. 2015 Sep;78:216-24. doi: 10.1016/j.bone.2015.05.016.
17. Hans D, Goertzen AL, Krieg MA, Leslie WD. Bone microarchitecture assessed by TBS predicts osteoporotic fractures independent of bone density: The Manitoba study. J Bone Miner Res. 2011 Nov;26(11):2762-9. doi: 10.1002/jbmr.499.
18. Chuang M-H, Chuang T-L, Koo M, Wang Y-F. Trabecular Bone Score Reflects Trabecular Microarchitecture Deterioration and Fragility Fracture in Female Adult Patients Receiving Glucocorticoid Therapy: A Pre-Post Controlled Study. Biomed Res Int. 2017;2017:4210217. doi: 10.1155/2017/4210217.
19. Pothuaud L, Barthe N, Krieg M-A, Mehsen N, Carceller P, Hans D. Evaluation of the potential use of trabecular bone score to complement bone mineral density in the diagnosis of osteoporosis: a preliminary spine BMD-matched, case-control study. J Clin Densitom. 2009 Apr-Jun;12(2):170-6. doi: 10.1016/j.jocd.2008.11.006.
20. Rabier B, Héraud A, Grand-Lenoir C, Winzen-
rieth R, Hans D. A multicentre, retrospective case-control study assessing the role of trabecular bone score (TBS) in menopausal Caucasian women with low areal bone mine–ral density (BMDa): Analysing the odds of vertebral fracture. Bone. 2010 Jan;46(1):176-81. doi: 10.1016/j.bone.2009.06.032.

/9-1.jpg)
/10-1.jpg)
/11-1.jpg)